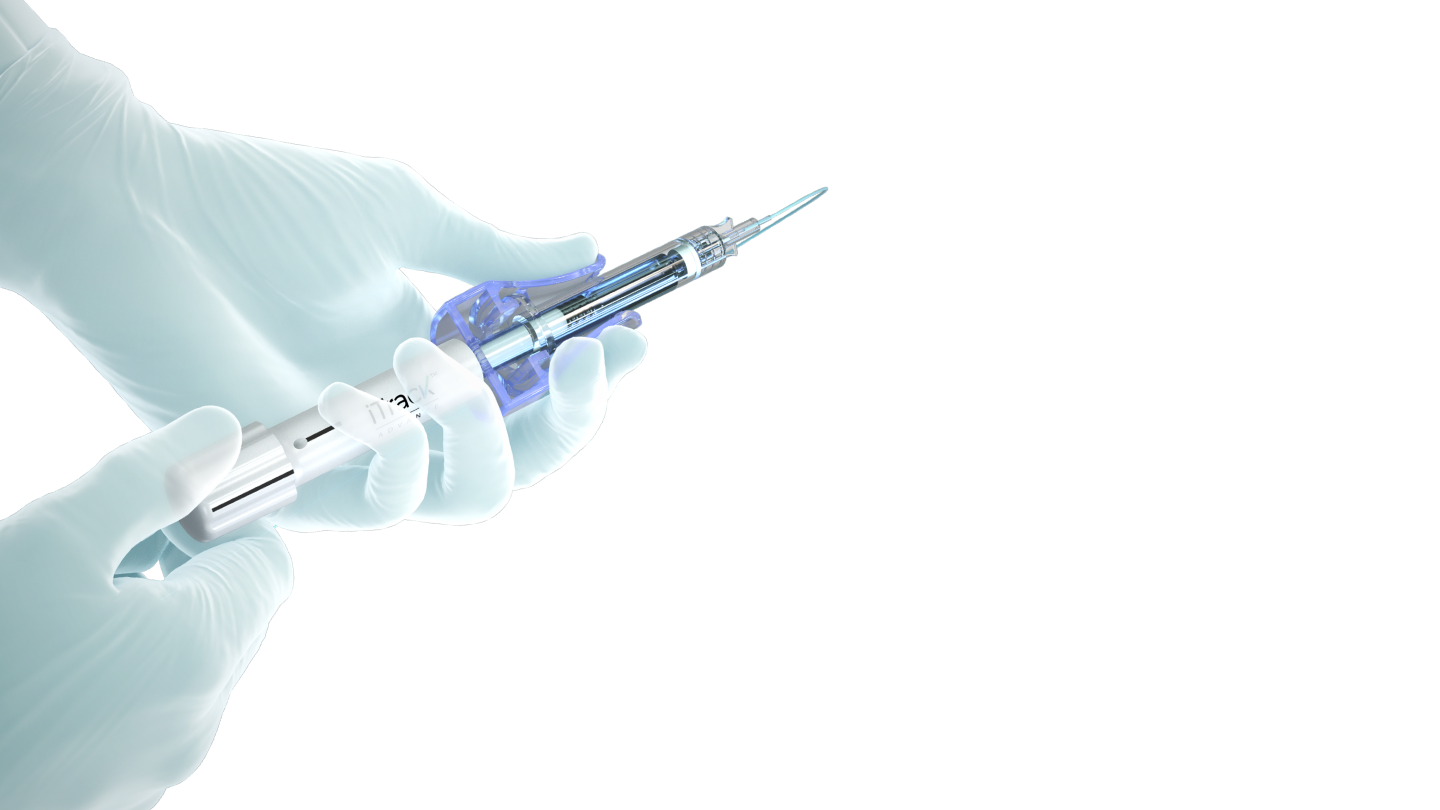
360º
Catheterization plus pressurized viscodilation across the full 360° of the conventional outflow pathway, supporting a comprehensive canaloplasty approach.

In-house manufacturing
Engineered and precision-crafted in our state-of-the-art facilities

Clinically proven iTrack™ technology
Built on the established iTrack™ platform, with clinical evidence supporting long-term effectiveness and real-world adoption. (1), (2)
1. Kerr, N., Lubeck, D., Barton, K., Aref, A. A., Cheng, J., Spaulding, J., Shoham-Hazon, N., Thomsen, S., Patel, S., Harasymowycz, P., Singh, H., Mercieca, K., & Ahmed, I. I. K. (2026). A Prospective, Real-World, Multicenter Study to Support the Role of Ab-Interno Canaloplasty in Glaucoma Management. American journal of ophthalmology, 284, 235–246.
2. Gallardo M. J. (2022). 36-Month Effectiveness of Ab-Interno Canaloplasty Standalone versus Combined with Cataract Surgery for the Treatment of Open-Angle Glaucoma. Ophthalmology. Glaucoma, 5(5), 476–482.